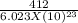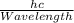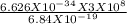Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

You know the right answer?
It takes 412. KJ/mol to break a carbon-hydrogen single bond. Calculate the maximum wavelength of lig...
Questions


Mathematics, 18.06.2020 17:57

Biology, 18.06.2020 17:57


Physics, 18.06.2020 17:57

Mathematics, 18.06.2020 17:57



Mathematics, 18.06.2020 17:57



Mathematics, 18.06.2020 17:57

Geography, 18.06.2020 17:57

English, 18.06.2020 17:57


Biology, 18.06.2020 17:57

Social Studies, 18.06.2020 17:57