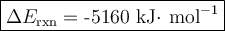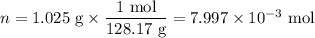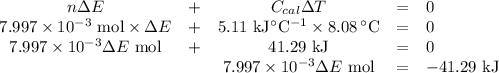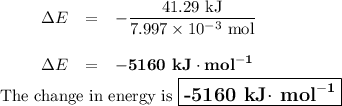Chemistry, 27.03.2020 21:01 usagimiller
Mothballs are composed primarily of the hydrocarbon naphthalene (C10H8). When 1.025 gof naphthalene is burned in a bomb calorimeter, the temperature rises from 24.25 ∘C to 32.33 ∘C. Find ΔErxn for the combustion of naphthalene. The heat capacity of the calorimeter, determined in a separate experiment, is 5.11kJ/∘C. Express the change in energy in kilojoules per mole to three significant figures.ΔErxn = kJ/mol

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Does the temperature affect the solubility of sugar and salt in water? if it does tell me like different temperatures with different solubilities so i can sketch down a graph
Answers: 2

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration?
Answers: 3

Chemistry, 21.06.2019 23:50
Working with si (metric) units for each of the following commonly used measurements, indicate its symbol. liter gram milliliter kilogram meter centigram milligram centimeter kilometer second millimeter milliseconds
Answers: 1
You know the right answer?
Mothballs are composed primarily of the hydrocarbon naphthalene (C10H8). When 1.025 gof naphthalene...
Questions



Mathematics, 26.07.2020 01:01














English, 26.07.2020 01:01