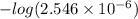Chemistry, 27.03.2020 16:43 clarkster112679
A chemist titrates 110.0 mL of a 0.7684 M methylamine (CH3NH2) sotion with 0.4469 M HNO3 solution at 25 °C. Calculate the pH at equivalence. The p K, of methylamine is 3.36 Round your answer to 2 decimal places Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of HNO3 solution added

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Does the temperature affect the solubility of sugar and salt in water? if it does tell me like different temperatures with different solubilities so i can sketch down a graph
Answers: 2

Chemistry, 22.06.2019 08:40
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3
You know the right answer?
A chemist titrates 110.0 mL of a 0.7684 M methylamine (CH3NH2) sotion with 0.4469 M HNO3 solution at...
Questions

Chemistry, 14.11.2020 07:20

Social Studies, 14.11.2020 07:20

History, 14.11.2020 07:20

SAT, 14.11.2020 07:20


Mathematics, 14.11.2020 07:20

Chemistry, 14.11.2020 07:20

English, 14.11.2020 07:20


Mathematics, 14.11.2020 07:20

Mathematics, 14.11.2020 07:20

Health, 14.11.2020 07:20

History, 14.11.2020 07:20

English, 14.11.2020 07:20


Mathematics, 14.11.2020 07:20

History, 14.11.2020 07:20


Physics, 14.11.2020 07:20

Mathematics, 14.11.2020 07:20

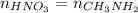
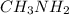 as follows.
as follows.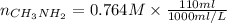
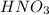 present will be calculated as follows.
present will be calculated as follows.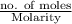
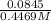
![[CH_{3}NH_{3}^{+}] = \frac{0.0845 mol}{0.2991 L}](/tpl/images/0567/3743/df2f2.png)
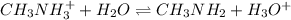
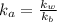
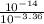
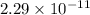
![[HNO_{3}] = \sqrt{k_{a}[CH_{3}NH_{3}^{+}]}](/tpl/images/0567/3743/a246e.png)
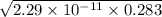
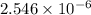
![-log [H_{3}O^{+}]](/tpl/images/0567/3743/df803.png)