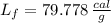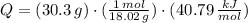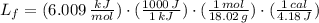Chemistry, 27.03.2020 04:29 dontcareanyonemo
The molar enthalpy of vaporization of water is 40.79 kJ/mol, and the molar enthalpy of fusion of ice is 6.009 kJ/mol. The molar mass of water is 18.02 g/mol. a. How much energy is absorbed when 30.3 g of liquid water boils? b. An energy unit often encountered is the calorie (4.18 J = 1 calorie). Determine the molar enthalpy of fusion of ice in calories per gram.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2

You know the right answer?
The molar enthalpy of vaporization of water is 40.79 kJ/mol, and the molar enthalpy of fusion of ice...
Questions

Mathematics, 27.05.2021 18:00


Mathematics, 27.05.2021 18:00







Mathematics, 27.05.2021 18:00




Mathematics, 27.05.2021 18:00



Biology, 27.05.2021 18:00

Mathematics, 27.05.2021 18:00


English, 27.05.2021 18:00

 , b)
, b)