
Chemistry, 26.03.2020 23:04 YOURKINGNESS7689
A lab technician mixes a 0.730 M solution of sodium bromide (NaBr) and water. The volume of the solution is 135 mililiters. What is the mass of dissolved sodium bromide in the solution?
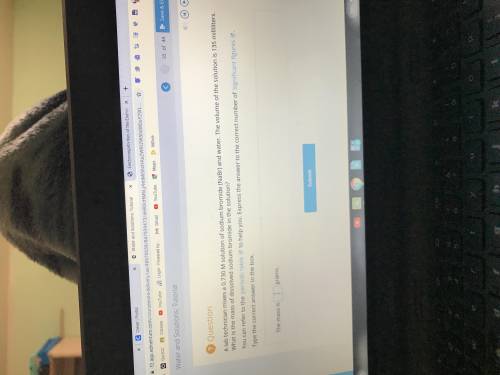

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:40
Water ionizes by the equation h2o(l)⇌h+(aq)+oh−(aq) the extent of the reaction is small in pure water and dilute aqueous solutions. this reaction creates the following relationship between [h+] and [oh−]: kw=[h+][oh−] keep in mind that, like all equilibrium constants, the value of kw changes with temperature.
Answers: 1

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 22:00
Plz ill give u brainliest which of the following steps is not likely to take place during cellular respiration? a.oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. d. energy is used up.
Answers: 3
You know the right answer?
A lab technician mixes a 0.730 M solution of sodium bromide (NaBr) and water. The volume of the solu...
Questions


Mathematics, 25.08.2019 01:00

Social Studies, 25.08.2019 01:00

Mathematics, 25.08.2019 01:00

Biology, 25.08.2019 01:00

Biology, 25.08.2019 01:00



Biology, 25.08.2019 01:00




Mathematics, 25.08.2019 01:00


History, 25.08.2019 01:00

Mathematics, 25.08.2019 01:00

Biology, 25.08.2019 01:00



Mathematics, 25.08.2019 01:00



