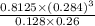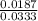Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:20
Which of the following statements is not true? • a. covalent compounds have low melting and boiling points. • ob. covalent bonds between atoms of a compound are relatively weak compared to bonds between molecules. • c. covalent bonds occur between nonmetals. • d. covalent compounds are often gases or liquids.
Answers: 2

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2
You know the right answer?
In a study of the conversion of methane to other fuels, a chemical engineer mixes gaseous CH4 and H2...
Questions


Mathematics, 07.05.2021 01:00

Mathematics, 07.05.2021 01:00

Mathematics, 07.05.2021 01:00




Mathematics, 07.05.2021 01:10

Mathematics, 07.05.2021 01:10

Mathematics, 07.05.2021 01:10

English, 07.05.2021 01:10

Engineering, 07.05.2021 01:10

Mathematics, 07.05.2021 01:10



Mathematics, 07.05.2021 01:10



Mathematics, 07.05.2021 01:10

![[H_{2}O]](/tpl/images/0566/0391/04475.png) at equilibrium is 0.561 M.
at equilibrium is 0.561 M.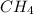 = 0.041 mol,
= 0.041 mol,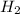 = 0.091 mol
= 0.091 mol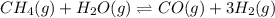
![\frac{[CO][H_{2}]^{3}}{[CH_{4}][H_{2}O]}](/tpl/images/0566/0391/7f129.png) ...... (1)
...... (1)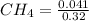 = 0.128 M,
= 0.128 M,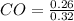 = 0.8125 M,
= 0.8125 M, 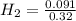 = 0.284 M
= 0.284 M![[H_{2}O] = \frac{[CO][H_{2}]^{3}}{[CH_{4}] \times K}](/tpl/images/0566/0391/7448a.png)