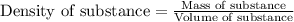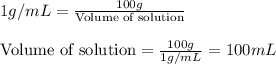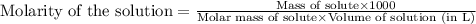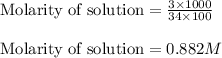Hydrogen peroxide, a disinfectant, contains 3.0% (w/w) hydrogen peroxide in water. This means there are 3.0 grams of hydrogen peroxide in every 100. grams of solution. Assuming this solution has a density of 1.00 g/mL, what is the molar concentration of this solution

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2


You know the right answer?
Hydrogen peroxide, a disinfectant, contains 3.0% (w/w) hydrogen peroxide in water. This means there...
Questions

English, 06.10.2019 22:20

English, 06.10.2019 22:20

Mathematics, 06.10.2019 22:20

English, 06.10.2019 22:20

Mathematics, 06.10.2019 22:20

English, 06.10.2019 22:20

Mathematics, 06.10.2019 22:20

History, 06.10.2019 22:20

History, 06.10.2019 22:20



Arts, 06.10.2019 22:30

Chemistry, 06.10.2019 22:30


Physics, 06.10.2019 22:30


Mathematics, 06.10.2019 22:30



Mathematics, 06.10.2019 22:30