
Chemistry, 26.03.2020 20:59 makaylashrout77
Acetylene (C2H2) gas is often used in welding torches because of the very high heat produced when it reacts with oxygen (O2) gas, producing carbon dioxide gas and water vapor. Calculate the moles of acetylene needed to produce 0.090mol of carbon dioxide. Be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Endeleev saw trends in the physical and chemical properties of elements when he organized them by
Answers: 2

Chemistry, 22.06.2019 04:30
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2
You know the right answer?
Acetylene (C2H2) gas is often used in welding torches because of the very high heat produced when it...
Questions

Physics, 24.09.2019 17:00







History, 24.09.2019 17:00


History, 24.09.2019 17:00

Mathematics, 24.09.2019 17:00

Mathematics, 24.09.2019 17:00






Chemistry, 24.09.2019 17:00


Business, 24.09.2019 17:00

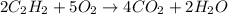
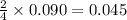 moles of acetylene
moles of acetylene

