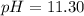Chemistry, 26.03.2020 20:36 gennhill14
A chemist titrates 100.0mL of a 0.3444M hydrocyanic acid HCN solution with 0.8414M KOH solution at 25°C . Calculate the pH at equivalence. The pKa of hydrocyanic acid is 9.21 . Round your answer to 2 decimal places.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 21:50
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l.s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2

Chemistry, 23.06.2019 04:20
The equation below shows a chemical reaction. a + b + heat —> c + d according to the law of conservation of energy, which statement is true? a. the reactants absorb heat because they have less energy than the products. b. the products release heat because they have more energy than the reactants. c. the reactants generate heat because they have more energy than the products. d. the products require heat to form because they have less energy than the reactants.
Answers: 1

Chemistry, 23.06.2019 09:30
How many moles of na2s2o3 are needed to react with 0.12mol of cl2? show work.
Answers: 1
You know the right answer?
A chemist titrates 100.0mL of a 0.3444M hydrocyanic acid HCN solution with 0.8414M KOH solution at 2...
Questions

Biology, 18.10.2019 04:10

Mathematics, 18.10.2019 04:10




Mathematics, 18.10.2019 04:10

Mathematics, 18.10.2019 04:10

Mathematics, 18.10.2019 04:10





Mathematics, 18.10.2019 04:10



Mathematics, 18.10.2019 04:10




Mathematics, 18.10.2019 04:10

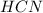 = 0.3444 M
= 0.3444 M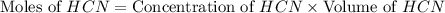
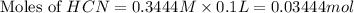
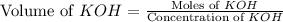
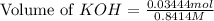
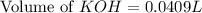
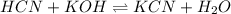
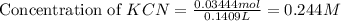
![pH=\frac{1}{2}[pK_w+pK_a+\log C]](/tpl/images/0565/6867/b44e5.png)
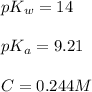
![pH=\frac{1}{2}[14+9.21+\log (0.244)]](/tpl/images/0565/6867/e6178.png)