
Chemistry, 25.03.2020 01:57 wedderman6049
At a certain temperature, the reaction 2NO + Cl2 ⇌ 2NOCl has an equilibrium constant Kc of 45.0. A chemist creates a mixture with the following initial concentrations: [NO] = 0.10 M, [Cl2] = 0.20 M, and [NOCl] = 0.30 M. What will happen as the reaction begins?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 00:30
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3

Chemistry, 22.06.2019 09:20
Sugar is dissolved in water. which is the solute? sugar neither both water
Answers: 1

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1
You know the right answer?
At a certain temperature, the reaction 2NO + Cl2 ⇌ 2NOCl has an equilibrium constant Kc of 45.0. A c...
Questions

Mathematics, 22.12.2020 01:00

Mathematics, 22.12.2020 01:00

Arts, 22.12.2020 01:00

World Languages, 22.12.2020 01:00

Mathematics, 22.12.2020 01:00

Computers and Technology, 22.12.2020 01:00

Mathematics, 22.12.2020 01:00

Social Studies, 22.12.2020 01:00






Arts, 22.12.2020 01:00

Mathematics, 22.12.2020 01:00





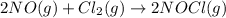 has an equilibrium constant Kc of 45.0. A chemist creates a mixture with the following initial concentrations: [NO] = 0.10 M, [Cl₂] = 0.20 M, and [NOCl] = 0.30 M. What will happen as the reaction begins?
has an equilibrium constant Kc of 45.0. A chemist creates a mixture with the following initial concentrations: [NO] = 0.10 M, [Cl₂] = 0.20 M, and [NOCl] = 0.30 M. What will happen as the reaction begins?![Q=\frac{[NOCl]^2}{[NO]^2[Cl_2]}](/tpl/images/0562/1779/afbe9.png)
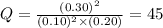
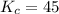
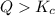 that means product > reactant. So, the reaction is reactant favored.
that means product > reactant. So, the reaction is reactant favored.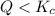 that means reactant > product. So, the reaction is product favored.
that means reactant > product. So, the reaction is product favored.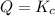 that means product = reactant. So, the reaction is in equilibrium.
that means product = reactant. So, the reaction is in equilibrium.

