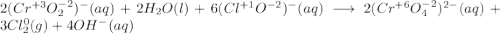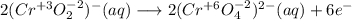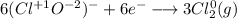Chemistry, 24.03.2020 20:58 grayjasmine46
Consider the following balanced redox reaction: 2CrO2-(aq) + 2H2O(l) + 6ClO-(aq) LaTeX: \longrightarrow⟶ 2CrO42-(aq) + 3Cl2(g) + 4OH-(aq) 1. Which species is being oxidized? 2. Which species is being reduced? 3. Which species is the oxidizing agent? 4. Which species is the reducing agent? 5. How many electrons are being transferred? Hint: If you were to balance this equation how many electrons would be in each half-reaction? That is how many electrons are transferred.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1


Chemistry, 23.06.2019 01:00
Reactions in cells take place at about a. 40°c b. 0° c. 100°c d. 60°c
Answers: 1
You know the right answer?
Consider the following balanced redox reaction: 2CrO2-(aq) + 2H2O(l) + 6ClO-(aq) LaTeX: \longrightar...
Questions

Geography, 25.01.2021 19:10






Mathematics, 25.01.2021 19:10

Mathematics, 25.01.2021 19:10

Mathematics, 25.01.2021 19:10

Arts, 25.01.2021 19:10


Mathematics, 25.01.2021 19:10

English, 25.01.2021 19:10



Biology, 25.01.2021 19:10



Mathematics, 25.01.2021 19:10

Mathematics, 25.01.2021 19:10