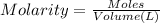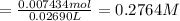Chemistry, 24.03.2020 19:14 roseemariehunter12
Initially a NaOH solution was standardized by titration with a sample of potassium hydrogenphthalate, KHC8H4O4, a monoprotic acid often used as a primary standard. A sample of pureKHC8H4O4 weighing 1.518 grams was dissolved in water and titrated with the NaOH solution. Toreach the equivalence point, 26.90 milliliters of base was required. Calculate the molarity of theNaOH solution. (Molecular weight: KHC8H4O4 = 204.2)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Anurse practitioner prepares an injection of promethazine, an antihistamine used to treat allergic rhinitis. if the stock bottle is labeled 25 mg/ml and the order is a dose of 11.0 mg , how many milliliters will the nurse draw up in the syringe?
Answers: 3

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 19:40
What is the wavelength of a 3*10^12 hz infrared wave a 3*10^20m b 1* 10^4m c 3*10^-3m d 1*10^-4 m
Answers: 1
You know the right answer?
Initially a NaOH solution was standardized by titration with a sample of potassium hydrogenphthalate...
Questions


Mathematics, 25.03.2020 05:42

Mathematics, 25.03.2020 05:42

Mathematics, 25.03.2020 05:42


Health, 25.03.2020 05:42


Computers and Technology, 25.03.2020 05:42

Geography, 25.03.2020 05:42

Mathematics, 25.03.2020 05:42

History, 25.03.2020 05:42


English, 25.03.2020 05:42

English, 25.03.2020 05:42





Mathematics, 25.03.2020 05:42

Mathematics, 25.03.2020 05:42

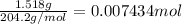
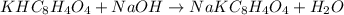
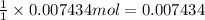 of NaOH
of NaOH