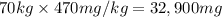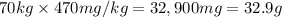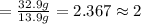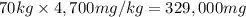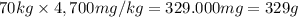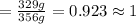Chemistry, 23.03.2020 22:50 rosiegirl313
Let the ED50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. If the ED50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz?If a bottle of beer contains about 13.9 g of ethanol, how many bottles of beer would a 70 kg party goer need to consume in order to have a 50% chance of getting a buzz?
If the lethal dose (LD50) for ethanol is 4,700 mg/kg, how many bottles of beer would a 70 kg party goer need to chug in order to have a 50% chance of dying?
A one liter bottle of Jack Daniels contains about 356 g of ethanol. How many 1.0 liter bottles of this whiskey would a 70 kg party goer need to chug in order to have a 50% chance of dying?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
George is a dalmatian puppy. describe what happens to light that allows you to see george’s black and white coat.
Answers: 1

Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 20:00
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1
You know the right answer?
Let the ED50 of a recreational drug be defined as the amount required for 50% of a test group to fee...
Questions

Mathematics, 27.09.2019 11:30


History, 27.09.2019 11:30

Biology, 27.09.2019 11:30

Mathematics, 27.09.2019 11:30

Mathematics, 27.09.2019 11:30

History, 27.09.2019 11:30




Biology, 27.09.2019 11:30

Mathematics, 27.09.2019 11:30

Chemistry, 27.09.2019 11:30

English, 27.09.2019 11:30

Biology, 27.09.2019 11:30


History, 27.09.2019 11:30

English, 27.09.2019 11:30

Social Studies, 27.09.2019 11:30

Physics, 27.09.2019 11:30