
Cerium (IV) ions are strong oxidizing agents in acid ic solution, oxidizing arsenio us acid to arsen ic acid accor ding to the following equa tion:
2Ce4+(aq)+H3AsO3(aq)+3H2O(l)→2Ce3+( aq)+H3AsO4(aq)+2H+(aq)
A sample of As2O3 weighing 0.217 g is dissolved in basic solution and then acidified to make H3AsO3. Its titration with a solution of acidic cerium{IV) sulfate requires 21.47 ml. Determine the original concentration of Ce^4+(aq) in the titrating solution

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Sex cells from female reproductive organ? 1) mitosis 2) fertilization 3) zygote 4) eggs 5) meiosis 6) sperm
Answers: 2

Chemistry, 22.06.2019 02:20
6. what does the symbol ah stand for? o one calorie given off by a reaction the specific heat of a substance the heat capacity of a substance the heat of reaction for a chemical reaction
Answers: 1

Chemistry, 22.06.2019 06:30
The minerals found in bones are deposited by living cells called
Answers: 1

Chemistry, 22.06.2019 11:10
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1
You know the right answer?
Cerium (IV) ions are strong oxidizing agents in acid ic solution, oxidizing arsenio us acid to arsen...
Questions

Mathematics, 26.12.2019 14:31



Social Studies, 26.12.2019 14:31



History, 26.12.2019 14:31



History, 26.12.2019 14:31




Mathematics, 26.12.2019 14:31


Mathematics, 26.12.2019 14:31



Mathematics, 26.12.2019 14:31

History, 26.12.2019 14:31

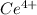 (aq) in the titrating solution.
(aq) in the titrating solution.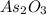 = 0.217 g
= 0.217 g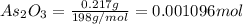
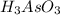 have 1 mole of As.
have 1 mole of As.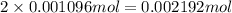 of
of 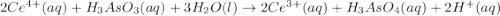
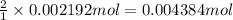 of cerium (IV) ions.
of cerium (IV) ions.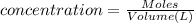
![[Ce^{4+}]=\frac{0.004384 mol}{0.02147 L}=0.2042 M](/tpl/images/0559/2089/fb855.png)


