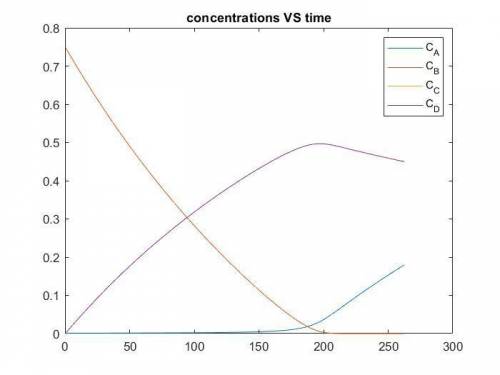
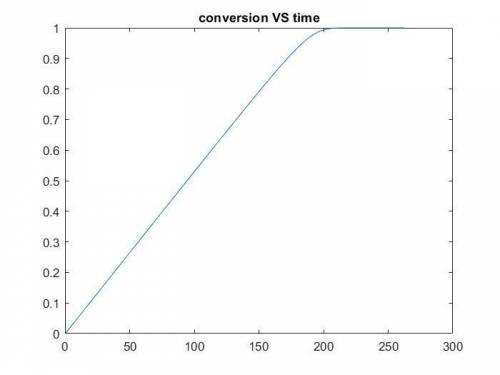
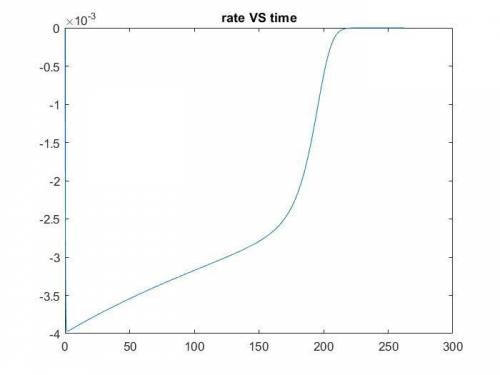
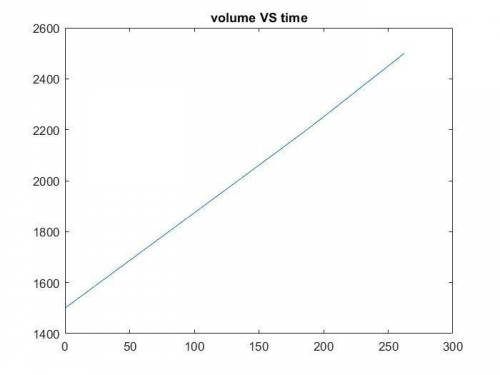
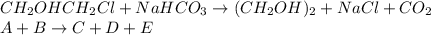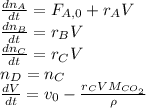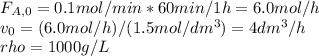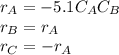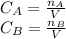The production of ethylene glycol from ethylene chlorohydrin and sodium bicarbonate ↑ is carried out in a semibatch reactor. A 1.5-molar solution of ethylene chlorohydrin is fed at a rate of 0.1 mole/minute to 1500 dm3 of a 0.75-molar solution of sodium bicarbonate. The reaction is elementary and carried out isothermally at 30°C where the specific reaction rate is 5.1 dm3/mol/h. Higher temperatures produce unwanted side reactions. The reactor can hold a maximum of 2500 dm3 of liquid. Assume constant density.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:10
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 09:00
Chen drew a diagram to compare the ways in which different organisms obtain nitrogen. which label belongs to the area marked z?
Answers: 3

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1
You know the right answer?
The production of ethylene glycol from ethylene chlorohydrin and sodium bicarbonate ↑ is carried out...
Questions



Social Studies, 08.07.2019 21:00

Chemistry, 08.07.2019 21:00

History, 08.07.2019 21:00

Mathematics, 08.07.2019 21:00

History, 08.07.2019 21:00


Mathematics, 08.07.2019 21:00

Mathematics, 08.07.2019 21:00



Biology, 08.07.2019 21:00


Biology, 08.07.2019 21:00

Biology, 08.07.2019 21:00

English, 08.07.2019 21:00


Mathematics, 08.07.2019 21:00

Computers and Technology, 08.07.2019 21:00