Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:50
What is the temperature of one mole of helium gas at stp?
Answers: 3


Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2

Chemistry, 23.06.2019 09:20
1) a. water molecule breaks up into hydrogen and oxygen on passing electricity. does this involve breaking intermolecular or intramolecular forces of attraction. explain b. on boiling water changes to water vapor. does this involve breaking intermolecular or intramolecular forces of attraction. explain methanol evaporates faster than water. contrast the intermolecular forces and the vapor pressures of methanol and water?
Answers: 2
You know the right answer?
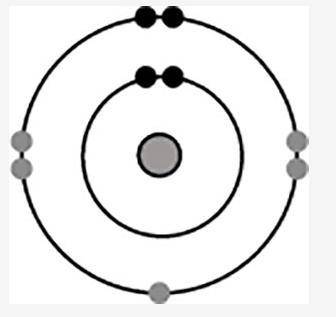
The diagram shows the electron configuration of an atom of an element for the electrons in the s and...
Questions

Mathematics, 16.10.2021 18:50


Mathematics, 16.10.2021 18:50


Mathematics, 16.10.2021 18:50


Spanish, 16.10.2021 18:50


Mathematics, 16.10.2021 18:50

Mathematics, 16.10.2021 18:50

Mathematics, 16.10.2021 18:50

Computers and Technology, 16.10.2021 18:50

Biology, 16.10.2021 18:50

Mathematics, 16.10.2021 18:50

Chemistry, 16.10.2021 18:50


Social Studies, 16.10.2021 18:50



English, 16.10.2021 18:50