Chemistry, 20.03.2020 07:19 valeriegarcia12
Nitric oxide (NO) can be formed from nitrogen, hydrogen and oxygen in two steps. In the first step, nitrogen and hydrogen react to form ammonia:
N2(g) + 3H2(g) →2NH3(g)
ΔH=−92.kJ
In the second step, ammonia and oxygen react to form nitric oxide and water:
4NH3(g) + 5O2(g) → 4NO(g) +6H2O(g)
ΔH=−905.kJ
Calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which of the following statements is true? a. elements in the last period are radioactive. b. atomic weight is the same as atomic mass. c. elements in the same group have the same number of electron shells. d. atomic number equals the number of neutrons in the nucleus of an atom.
Answers: 1

Chemistry, 22.06.2019 01:00
Agas occupies 475 cm^3 at 313k. find its volume at 367k. you must show all of your work to receive credit. be sure to identify which of the gas laws you will be using
Answers: 2


Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
You know the right answer?
Nitric oxide (NO) can be formed from nitrogen, hydrogen and oxygen in two steps. In the first step,...
Questions



English, 27.10.2020 16:00


Mathematics, 27.10.2020 16:00




Physics, 27.10.2020 16:00


English, 27.10.2020 16:00

Mathematics, 27.10.2020 16:00

Mathematics, 27.10.2020 16:00

Mathematics, 27.10.2020 16:00


Advanced Placement (AP), 27.10.2020 16:00

Mathematics, 27.10.2020 16:00



Computers and Technology, 27.10.2020 16:00

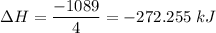
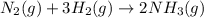 (1)
(1)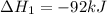
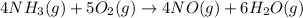 (2)
(2)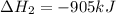
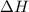 for the following reaction i.e,
for the following reaction i.e,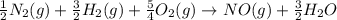
 for the reaction will be:
for the reaction will be: