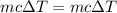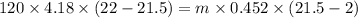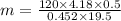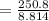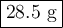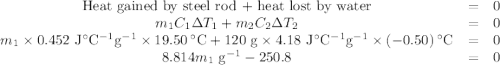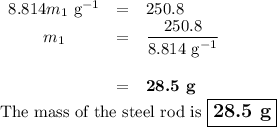Chemistry, 20.03.2020 05:58 soonerlady19
A volume of 120 mL of H2O is initially at room temperature (22.00 ∘C ). A chilled steel rod at 2.00 ∘C ∘C is placed in the water. If the final temperature of the system is 21.50 ∘C ∘C , what is the mass of the steel bar? Use the following values: specific heat of water = 4.18 J/(g⋅∘C)J/(g⋅∘C) specific heat of steel = 0.452 J/(g⋅∘C)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2


Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1
You know the right answer?
A volume of 120 mL of H2O is initially at room temperature (22.00 ∘C ). A chilled steel rod at 2.00...
Questions

Mathematics, 16.01.2020 01:31



Computers and Technology, 16.01.2020 01:31

Computers and Technology, 16.01.2020 01:31


Mathematics, 16.01.2020 01:31

Chemistry, 16.01.2020 01:31

Mathematics, 16.01.2020 01:31


Chemistry, 16.01.2020 01:31

English, 16.01.2020 01:31





Engineering, 16.01.2020 01:31