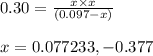For the reaction POCl3(g) ⇀↽ POCl(g) + Cl2(g) Kc = 0.30. An initial 0.3 moles of POCl3 are placed in a 3.1 L container with initial concentrations of POCl and Cl2 equal to zero. What is the final concentration of POCl3? 1. final concentration = 0.281774 M 2. final concentration = 0.077 M 3. final concentration = 0.185 M 4. final concentration = 0.039534 M 5. final concentration = 0.019767 M

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
The first element on the periodic table of elements is carbon. a. true b. false
Answers: 2

Chemistry, 22.06.2019 02:10
3.) for each of the following compounds, draw the major organic product of reaction with hcl or naoh and circle whether the starting materials and products will be more soluble in organic solvent or water benzoic acid + hcl: benzoic acid + naoh: oh benzoic acid water/organic water organic fluorenone hс: fluorenone + naoh: fluorenone water/organic water/organic веnzocaine + hci: benzocaine + n»oh: h2n benzocaine water/organic water organic o=
Answers: 3

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

You know the right answer?
For the reaction POCl3(g) ⇀↽ POCl(g) + Cl2(g) Kc = 0.30. An initial 0.3 moles of POCl3 are placed in...
Questions


English, 03.07.2019 06:00

Biology, 03.07.2019 06:00


Mathematics, 03.07.2019 06:00




History, 03.07.2019 06:00

History, 03.07.2019 06:00

Mathematics, 03.07.2019 06:00

Mathematics, 03.07.2019 06:00

Mathematics, 03.07.2019 06:00


Mathematics, 03.07.2019 06:00



History, 03.07.2019 06:00

Biology, 03.07.2019 06:00

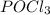 is 0.019767 M
is 0.019767 M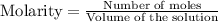
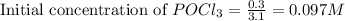
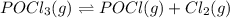
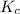 for above equation follows:
for above equation follows:![K_c=\frac{[POCl][Cl_2]}{[POCl_3]}](/tpl/images/0555/1089/0f7c6.png)