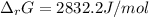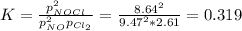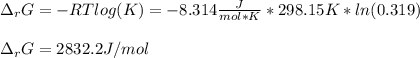Chemistry, 20.03.2020 00:06 nguyenhoangthienkim0
A chemist fills a reaction vessel with 9.47 atm nitrogen monoxide (NO) gas, 2.61 atm chlorine (C12) gas, and 8.64 atm nitrosyl chloride (NOCI) gas at a temperature of 25.0°C. Under these conditions, calculate the reaction free energy AG for the following chemical reaction:
2NO(g) + Cl2(g) <=> 2NOCI (g)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2

Chemistry, 23.06.2019 02:00
To calculate the molarity of a solution, you need to know the moles of solute and the
Answers: 2
You know the right answer?
A chemist fills a reaction vessel with 9.47 atm nitrogen monoxide (NO) gas, 2.61 atm chlorine (C12)...
Questions


Geography, 19.09.2019 08:30

History, 19.09.2019 08:30


Health, 19.09.2019 08:30




History, 19.09.2019 08:30


Geography, 19.09.2019 08:30



Social Studies, 19.09.2019 08:30






Health, 19.09.2019 08:30