
For the reaction CO2(g) + H2O(ℓ) + CaCl2(aq) ←→ CaCO3(s) + HCl(aq) K = 4.8 the concentrations of CO2, H2O, CaCl2 and HCl are 0.377 M, 55.4 M, 0.652 M and 30.2 M, respectively. What will happen in order for the system to reach equilibrium? 1. The reaction will shift to the right. 2. Nothing will occur. 3. The reaction will shift to the left. 4. Not enough information is given.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2


Chemistry, 22.06.2019 17:30
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2
You know the right answer?
For the reaction CO2(g) + H2O(ℓ) + CaCl2(aq) ←→ CaCO3(s) + HCl(aq) K = 4.8 the concentrations of CO2...
Questions

History, 27.06.2019 09:30



Mathematics, 27.06.2019 09:30

Biology, 27.06.2019 09:30




Mathematics, 27.06.2019 09:30


Mathematics, 27.06.2019 09:30



Physics, 27.06.2019 09:30

Biology, 27.06.2019 09:30





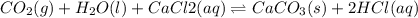
![K=\frac{[HCl]^2}{[CaCl_2]}](/tpl/images/0554/8954/74ccf.png)
![Q=\frac{[HCl]^2}{[CaCl_2]}=\frac{ 30.2^2 }{0.652 }=1398.8](/tpl/images/0554/8954/f4b79.png)


