
Chemistry, 19.03.2020 22:45 anthonylopez1
When aluminum, AlAl, metal is dipped in an aqueous solution of hydrochloric acid, HClHCl, hydrogen gas, H2H2, is produced with the formation of an aluminum chloride, AlCl3AlCl3, solution. Write the balanced chemical equation showing the phases of reactants and products.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3
You know the right answer?
When aluminum, AlAl, metal is dipped in an aqueous solution of hydrochloric acid, HClHCl, hydrogen g...
Questions


History, 18.03.2021 01:50

Advanced Placement (AP), 18.03.2021 01:50

Physics, 18.03.2021 01:50


Mathematics, 18.03.2021 01:50


World Languages, 18.03.2021 01:50


Biology, 18.03.2021 01:50


Mathematics, 18.03.2021 01:50


History, 18.03.2021 01:50



Mathematics, 18.03.2021 01:50

Mathematics, 18.03.2021 01:50

Mathematics, 18.03.2021 01:50

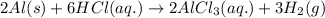
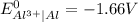 ).
).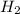 is present at the intermediate position in electrochemical series with a standard reduction potential of 0 V (
is present at the intermediate position in electrochemical series with a standard reduction potential of 0 V (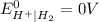 ).So, when Al is dipped in aqueous solution of HCl, Al is readily oxidized to produce
).So, when Al is dipped in aqueous solution of HCl, Al is readily oxidized to produce 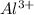 whereas
whereas 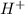 is reduced to
is reduced to 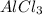 and gaseous
and gaseous 


