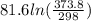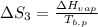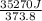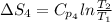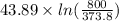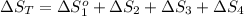The standard entropy of liquid methanol at 298K is 126.8 J/K-mol and its heat capacity is 81.6 J/K-mol. Methanol boils at 337K with an enthalpy of vaporization of 35.270 kJ/mol at that temperature. The heat capacity of the vapor is 43.9 J/K-mol.__Calculate the entropy of one mole of methanol vapor at 800 K.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Which compound contains both ionic and covalent bonds? a) hbr b)cbr4 c)nabr d) naoh
Answers: 2

Chemistry, 22.06.2019 18:50
At stp, which substance is the best conductor of electricity? a. nitrogen b. neon c. sulfur d. silver
Answers: 1

Chemistry, 23.06.2019 00:00
In an exothermic reaction, energy may be released to the surroundings in the form of question 4 options: heat light thermal all of the above
Answers: 3

Chemistry, 23.06.2019 03:00
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 1
You know the right answer?
The standard entropy of liquid methanol at 298K is 126.8 J/K-mol and its heat capacity is 81.6 J/K-m...
Questions

English, 13.12.2019 20:31





English, 13.12.2019 20:31

Mathematics, 13.12.2019 20:31


Arts, 13.12.2019 20:31


Mathematics, 13.12.2019 20:31


History, 13.12.2019 20:31


Mathematics, 13.12.2019 20:31

Mathematics, 13.12.2019 20:31

English, 13.12.2019 20:31


History, 13.12.2019 20:31

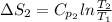 J/K mol
J/K mol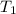 = 298 K,
= 298 K, 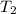 = 373.8 K
= 373.8 K