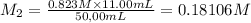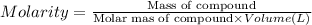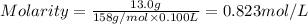Chemistry, 19.03.2020 20:56 Uhmjujiooo45701
By pipet, 11.00 mL of a 0.823 MM stock solution of potassium permanganate (KMnO4) was transferred to a 50.00-mL volumetric flask and diluted to the calibration mark. Determine the molarity of the resulting solution. A stock solution of potassium permanganate (KMnO4) was prepared by dissolving 13.0g KMnO4 with DI H2O in a 100.00-mL volumetric flask and diluting to the calibration mark. Determine the molarity of the solution Molarity= O.822 M

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1


Chemistry, 23.06.2019 01:30
Which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1

Chemistry, 23.06.2019 02:00
What is the difference between a substance "getting wet" and "being dissolved" in a liquid at the particulate level?
Answers: 3
You know the right answer?
By pipet, 11.00 mL of a 0.823 MM stock solution of potassium permanganate (KMnO4) was transferred to...
Questions

English, 25.08.2020 23:01

Mathematics, 25.08.2020 23:01





Physics, 25.08.2020 23:01


Mathematics, 25.08.2020 23:01




English, 25.08.2020 23:01


Mathematics, 25.08.2020 23:01



Engineering, 25.08.2020 23:01


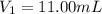
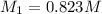
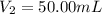
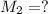
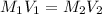 ( dilution )
( dilution )