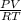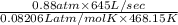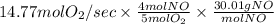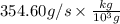Chemistry, 19.03.2020 08:01 leomessifanboy678
Suppose a chemical engineer studying a new catalyst for the oxidation of ammonia reaction finds that liters per second of dioxygen are consumed when the reaction is run at and . Calculate the rate at which nitrogen monoxide is being produced. Give your answer in kilograms per second. Round your answer to significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 14:30
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1

Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3

Chemistry, 23.06.2019 02:00
What are fossils of organisms that existed over a wide area but only for a limited time period called?
Answers: 2
You know the right answer?
Suppose a chemical engineer studying a new catalyst for the oxidation of ammonia reaction finds that...
Questions

Mathematics, 29.06.2019 21:40


English, 29.06.2019 21:40


Advanced Placement (AP), 29.06.2019 21:40



History, 29.06.2019 21:40


Mathematics, 29.06.2019 21:40


Mathematics, 29.06.2019 21:40


Chemistry, 29.06.2019 21:40

Chemistry, 29.06.2019 21:40

English, 29.06.2019 21:40

Mathematics, 29.06.2019 21:40



History, 29.06.2019 21:40

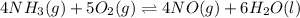
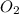 per second consumed is as follows.
per second consumed is as follows.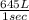
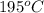 (468.15 K) and pressure 0.88 atm. Hence, moles of consumption of
(468.15 K) and pressure 0.88 atm. Hence, moles of consumption of