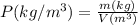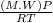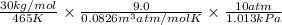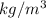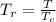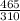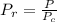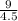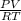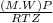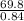Chemistry, 19.03.2020 07:58 kelseiroll9759
A certain gas has a molecular weight of 30.0, a critical temperature of 310 K, and a critical pressure of 4.5 MPa. Calculate the density in kg / m 3 kg/m3 of this gas at 465 K and 9.0 MPa (a) if the gas is ideal and (b) if the gas obeys the law of corresponding states

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3
You know the right answer?
A certain gas has a molecular weight of 30.0, a critical temperature of 310 K, and a critical pressu...
Questions

Mathematics, 05.10.2019 10:30

Engineering, 05.10.2019 10:30

History, 05.10.2019 10:30


Health, 05.10.2019 10:30


Social Studies, 05.10.2019 10:30

Engineering, 05.10.2019 10:30





Engineering, 05.10.2019 10:30

Engineering, 05.10.2019 10:30

History, 05.10.2019 10:30

Computers and Technology, 05.10.2019 10:30

Computers and Technology, 05.10.2019 10:30

Mathematics, 05.10.2019 10:30

History, 05.10.2019 10:30

Computers and Technology, 05.10.2019 10:30