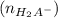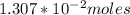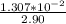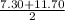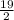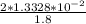Chemistry, 19.03.2020 07:14 yesseniaroman21
In the titration of 238.0 mL of a 5.60×10-2 M solution of acid H3A (Ka1 = 1.0×10-3, Ka2 = 5.0×10-8, Ka3 = 2.0×10-12), calculate the volume of 2.90 M NaOH required to reach the following pH values. pH = 9.50 Volume required = mL pH = 4.70 Volume required = mL

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1

Chemistry, 22.06.2019 22:50
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3

Chemistry, 23.06.2019 06:00
If you try to move a piano and are unable to move it, did you perform any work in the scientific sense of the word? yes? or no? this question is worth 20 points! let it be correct!
Answers: 1
You know the right answer?
In the titration of 238.0 mL of a 5.60×10-2 M solution of acid H3A (Ka1 = 1.0×10-3, Ka2 = 5.0×10-8,...
Questions

Mathematics, 07.04.2020 11:07


Mathematics, 07.04.2020 11:08

History, 07.04.2020 11:10





History, 07.04.2020 11:53

Mathematics, 07.04.2020 11:53

Mathematics, 07.04.2020 11:53



Mathematics, 07.04.2020 11:53

Social Studies, 07.04.2020 11:53

Mathematics, 07.04.2020 11:54




History, 07.04.2020 11:55

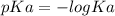
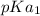 ; we have
; we have 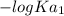
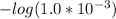
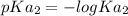
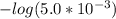
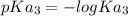
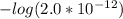
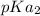 and
and 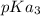 .
.![[H_3O^+]](/tpl/images/0553/6901/6cfd2.png) = pH = 4.70
= pH = 4.70![[H_3O^+] = 10^{-4.70}](/tpl/images/0553/6901/8c265.png)
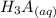

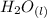 ⇄
⇄ 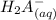
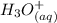
![Ka_1= \frac{[H_2A^-][H_3O^+]}{[H_3A]}](/tpl/images/0553/6901/4cf6a.png)
![1.0*10^{-3}= \frac{[H_2A^-][H_3O^+]}{[H_3A]}](/tpl/images/0553/6901/72bac.png)
![\frac{1.0*10^{-3}}{[H_3O^+]}= \frac{[H_2A^-]}{[H_3A]}](/tpl/images/0553/6901/eb284.png)
![\frac{1.0*10^{-3}}{[10^{-4.70}]}= \frac{[H_2A^-]}{[H_3A]}](/tpl/images/0553/6901/c148a.png)
![\frac{[H_2A^-]}{[H_3A]}= 10^{1.7}](/tpl/images/0553/6901/e4f78.png)
![\frac{[H_2A^-]}{[H_3A]}= 50.12](/tpl/images/0553/6901/480fb.png)
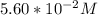 solution is given:
solution is given: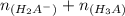 =
= 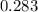
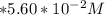
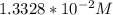
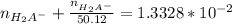
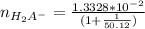
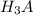 to
to 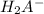 i.e
i.e