Chemistry, 19.03.2020 01:58 tristasbarker03
Calculate the volume of carbon dioxide at 20.0°C and 0.941 atm produced from the complete combustion of 4.00 kg of methane. Compare your result with the volume of CO2 produced from the complete combustion of 4.00 kg of propane (C3H8).

Answers: 3


Another question on Chemistry

Chemistry, 23.06.2019 00:00
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3

Chemistry, 23.06.2019 03:00
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2

Chemistry, 23.06.2019 04:31
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1

Chemistry, 23.06.2019 08:40
Calculate the number of grams of sodium in 3.00 g of each sodium-containing food additive.
Answers: 3
You know the right answer?
Calculate the volume of carbon dioxide at 20.0°C and 0.941 atm produced from the complete combustion...
Questions


English, 30.08.2019 08:50




Social Studies, 30.08.2019 08:50


Mathematics, 30.08.2019 08:50


History, 30.08.2019 08:50



Mathematics, 30.08.2019 08:50

English, 30.08.2019 08:50

Mathematics, 30.08.2019 08:50




Mathematics, 30.08.2019 08:50

Mathematics, 30.08.2019 08:50

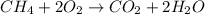
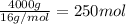
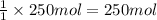 of carbon dioxide gas
of carbon dioxide gas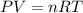 (Ideal gas equation)
(Ideal gas equation)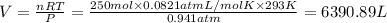
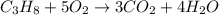
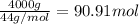
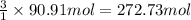 of carbon dioxide gas
of carbon dioxide gas