
Chemistry, 18.03.2020 22:30 pumaben2864
In the laboratory you dissolve 22.8 g of chromium(III) acetate in a volumetric flask and add water to a total volume of 250 mL. What is the molarity of the solution? M. What is the concentration of the chromium(III) cation? M. What is the concentration of the acetate anion?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 23.06.2019 01:00
What type of chemical bond is formed between two atoms of bromine 1. metallic 2. hydrogen 3. ionic 4. covalent
Answers: 1

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
In the laboratory you dissolve 22.8 g of chromium(III) acetate in a volumetric flask and add water t...
Questions

Mathematics, 19.10.2019 02:00


Physics, 19.10.2019 02:00

Mathematics, 19.10.2019 02:00





History, 19.10.2019 02:00

Physics, 19.10.2019 02:00


Mathematics, 19.10.2019 02:00

Biology, 19.10.2019 02:00

Physics, 19.10.2019 02:00

Mathematics, 19.10.2019 02:00

Social Studies, 19.10.2019 02:00




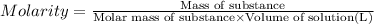
![[Cr(CH_3COO)_3]=\frac{22.8 g}{229 g/mol\times 0.250 L}=0.3982 M](/tpl/images/0552/7202/c3854.png)
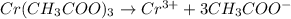
![[Cr^{3+}]=[Cr(CH_3COO)_3]=0.3982 M](/tpl/images/0552/7202/a4c9c.png)
![[CH_3COO^-]=3\times [Cr(CH_3COO)_3]=3\times 0.3892 M=1.1946 M](/tpl/images/0552/7202/97a2c.png)


