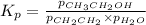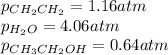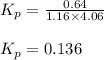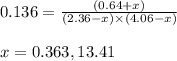Chemistry, 18.03.2020 22:08 kellinvagneur
While ethanol is produced naturally by fermentation, e. g. in beer- and wine-making, industrially it is synthesized by reacting ethylene with water vapor at elevated temperatures. A chemical engineer studying this reaction fills a flask with of ethylene gas and of water vapor. When the mixture has come to equilibrium she determines that it contains of ethylene gas and of water vapor. The engineer then adds another of ethylene, and allows the mixture to come to equilibrium again. Calculate the pressure of ethanol after equilibrium is reached the second time. Round your answer to significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 10:30
Rocks, as they are compressed, begin forming mountains above the earth's surface when two continental plates converge. the continental crust increases in depth as the mountains grow above. the himalayan mountains formed at a convergent plate boundary in this manner. the rocks are smashed together causing them to due to the intense heat and pressure from the colliding plates and eventually forming rock. a) melt; igneous b) layer; sedimentary c) recrystallize; metamorphic d) melt into the earth's interior; metamorphic
Answers: 1


Chemistry, 22.06.2019 15:30
The identities of substances are the same before and after which type of change
Answers: 1
You know the right answer?
While ethanol is produced naturally by fermentation, e. g. in beer- and wine-making, industrially it...
Questions

Mathematics, 06.12.2021 06:40



Chemistry, 06.12.2021 06:40




English, 06.12.2021 06:40




History, 06.12.2021 06:40

Social Studies, 06.12.2021 06:40

Mathematics, 06.12.2021 06:40

Mathematics, 06.12.2021 06:40


Biology, 06.12.2021 06:40



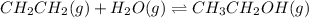
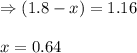
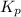 for above equation follows:
for above equation follows: