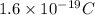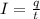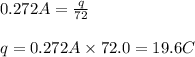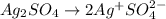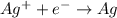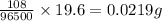Electroplating is a way to coat a complex metal object with a very thin (and hence inexpensive) layer of a precious metal, such as silver or gold. In essence the metal object is made the cathode of an electrolytic cell in which the precious metal cations are dissolved in aqueous solution. Suppose a current of is passed through an electroplating cell with an aqueous solution of in the cathode compartment for seconds. Calculate the mass of pure silver deposited on a metal object made into the cathode of the cell. Be sure your answer has a unit symbol and the correct number of significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Ineed someone to see if my answers are correct! if any are wrong let me know what the correct answers would be and how to get that answer! 1. how many moles of sodium chloride are in 28 grams od nacl? a. 265 mole naclb. 856 mole naclc. 479 mole of nacld. 1.2 mole nacl < my choice2. 734 grams of lithium sulfate (li2so4) are dissolved to make 2500 ml of solution what is rhe molaratiy? a. 2.67 mb. 4.56 mc. 3.89 m < my choiced. 1.78 m3. how many grams of cacl2 would be dissolved in 3.0 l of a 0.50 m solution of cacl2? a. 250 g cacl2 b. 166.5 g cacl2c. 113.65 g cacl2d. 98 g cacl2 < my choice4. suppose you had 58.44 g of nacl and you dissolved it in exactly 2.00 liters. the molarity if the solution would be 0.5 mtrue < my choicefalse 5. i would need 22g of naoh to make a 3.0 m solution using 250 ml of solvent.true < my choicefalse6. identify the solute: you have a .0195 m solution made from using 6.5 g of solute and 3 l of solvent. identify the solute by solving for molar weight.a. the solute is nacl because the molar weight is 58.43 g/mol < my choiceb. the solute is h2so4 because the molar weight is 98.06 g/molc. the solute is cacl2 because the molar weight is 111.11 g/mol
Answers: 1

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2


Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2
You know the right answer?
Electroplating is a way to coat a complex metal object with a very thin (and hence inexpensive) laye...
Questions

Arts, 22.04.2020 00:23



History, 22.04.2020 00:23

Mathematics, 22.04.2020 00:23





English, 22.04.2020 00:23

Biology, 22.04.2020 00:23

Mathematics, 22.04.2020 00:23

Geography, 22.04.2020 00:23

Mathematics, 22.04.2020 00:23

Mathematics, 22.04.2020 00:23

Biology, 22.04.2020 00:23


English, 22.04.2020 00:23


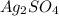 in the cathode compartment for 72 seconds. Calculate the mass of pure silver deposited on a metal object made into the cathode of the cell. Be sure your answer has a unit symbol and the correct number of significant digits.
in the cathode compartment for 72 seconds. Calculate the mass of pure silver deposited on a metal object made into the cathode of the cell. Be sure your answer has a unit symbol and the correct number of significant digits.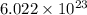 number of particles.
number of particles.