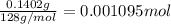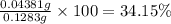Chemistry, 18.03.2020 21:26 Carlosanddana123
You are given a sample of limestone, which is mostly CaCO3, to determine the mass percentage of Ca in the rock. You dissolve the limestone in hydrochloric acid, which gives a solution of calcium chloride. Then you precipitate the calcium ion in solution by adding sodium oxalate, Na2C2O4. The precipitate is calcium oxalate, CaC2O4. You find that a sample of limestone weighing 128.3 mg gives 140.2 mg of CaC2O4. What is the mass percentage of calcium in the limestone

Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 22:20
Which of the following statements is false regarding aromaticity? a. the compound must be cyclic b. the compound must be fully conjugated c. the compound must be planar d.the number of electrons in the pi system must satisfy the hückel 4n+2 rule e. the compound must have a neutral charge
Answers: 2

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3

Chemistry, 23.06.2019 05:00
Select the statement that describe chemical properties a. antacid tablets neutralize stomach acid b. helium is the lightest monatomic element c. water freezes at 0 celsius d. mercury is liquid at room temperature
Answers: 3
You know the right answer?
You are given a sample of limestone, which is mostly CaCO3, to determine the mass percentage of Ca i...
Questions




Mathematics, 06.01.2020 04:31



Mathematics, 06.01.2020 04:31

Geography, 06.01.2020 04:31

Biology, 06.01.2020 04:31

Mathematics, 06.01.2020 04:31

Mathematics, 06.01.2020 04:31





Mathematics, 06.01.2020 04:31



History, 06.01.2020 04:31

English, 06.01.2020 04:31