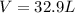Chemistry, 18.03.2020 20:05 Jadaaaaaaan
Determine the volume of SO2 (at STP) formed from the reaction of 96.7 g of FeS2 and 55.0 L of O2 (at 398 K and 1.20 atm). The molar mass of FeS2 is 119.99 g/mol. 4 FeS2(s) + 11 O2(g) → 2 Fe2O3(s) + 8 SO2(g)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2

Chemistry, 22.06.2019 05:30
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1
You know the right answer?
Determine the volume of SO2 (at STP) formed from the reaction of 96.7 g of FeS2 and 55.0 L of O2 (at...
Questions


Biology, 17.06.2021 19:40

Mathematics, 17.06.2021 19:40




Chemistry, 17.06.2021 19:40







Mathematics, 17.06.2021 19:40

Mathematics, 17.06.2021 19:40

Mathematics, 17.06.2021 19:40




Mathematics, 17.06.2021 19:40

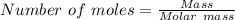


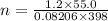
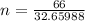
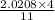 moles of FeS₂ will react with 2.0208 moles of oxygen to produce
moles of FeS₂ will react with 2.0208 moles of oxygen to produce 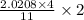 moles of SO₂
moles of SO₂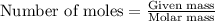
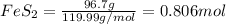

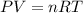

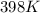


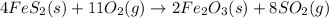
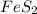
 moles of
moles of 
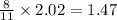 moles of
moles of 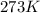 (at STP)
(at STP)