
Find the pH of each mixture of acids. Acid Ionization Constants (Ka) for Some Monoprotic Weak Acids at 25 ∘C Acid Formula Ka Benzoic acid HC7H5O2 6.5×10−5 Hydrofluoric acid HF 6.8×10−4 Phenol HC6H5O 1.3×10−10 Formic acid HCHO2 1.8×10−4 Hypochlorous acid HClO 2.9×10−8

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Drive down any three characteristic of modern periodic table
Answers: 1

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 22.06.2019 10:00
How many mmols of tris-hcl are there in 100 ml of a 100 mm tris-hcl buffer solution at ph 8.1? note that the 100 mm refers to the sum of tris and tris-hcl concentrations?
Answers: 3

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1
You know the right answer?
Find the pH of each mixture of acids. Acid Ionization Constants (Ka) for Some Monoprotic Weak Acids...
Questions

English, 03.12.2020 04:00



History, 03.12.2020 04:00

Chemistry, 03.12.2020 04:00


English, 03.12.2020 04:00


Mathematics, 03.12.2020 04:00

Mathematics, 03.12.2020 04:00




French, 03.12.2020 04:00

Mathematics, 03.12.2020 04:00


Computers and Technology, 03.12.2020 04:00


Geography, 03.12.2020 04:00

English, 03.12.2020 04:00

![[H^+] = \sqrt{K_2C}](/tpl/images/0551/1514/821c9.png)
![[H^+] = \sqrt{6.5*10^{-5}*0.185M}](/tpl/images/0551/1514/28253.png)
![[H^+] =0.0035M](/tpl/images/0551/1514/0669f.png)
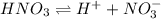
![[H^+]](/tpl/images/0551/1514/07acb.png) = 0.070 M
= 0.070 M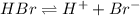
![[H^+] = 0.025 M](/tpl/images/0551/1514/a4539.png)
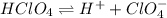
![[H^+] = 0.020 M](/tpl/images/0551/1514/f72cd.png)
![[H^+] = 0.020 M + 0.025 M](/tpl/images/0551/1514/f46a7.png)
![[H^+] = 0.045 M](/tpl/images/0551/1514/4bd75.png)
![[H^+] = \sqrt{K_{a1}C_1+K_{a2}C_2}](/tpl/images/0551/1514/9f22b.png)
![[H^+] = \sqrt{6.8*10^{-4}*0.095 +1,3*10^{-10}*0.230](/tpl/images/0551/1514/a064b.png)
![[H^+] = 0.008037 M](/tpl/images/0551/1514/33641.png)


