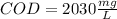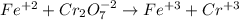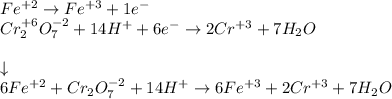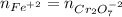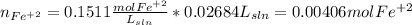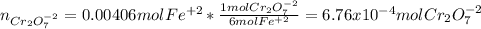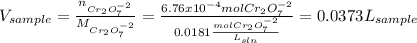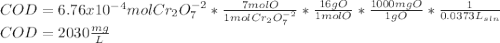Your standard iron solution is 0.1511 M Fe(II), your dichromate solution is 0.0181 M dichromate, and it took 26.84 mL of your standard iron solution to titrate the excess dichromate in your unknown. What is the chemical oxygen demand of the sample, in units of mg O2/L? (Report your answer with 4 sig figs)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 04:00
The continuous release of nuclear energy caused when one fission reaction triggered more nuclear reactions is a
Answers: 3

Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1

Chemistry, 22.06.2019 18:40
What is one real world example of a colligative property?
Answers: 2
You know the right answer?
Your standard iron solution is 0.1511 M Fe(II), your dichromate solution is 0.0181 M dichromate, and...
Questions

Mathematics, 03.11.2020 19:10


Mathematics, 03.11.2020 19:10




Physics, 03.11.2020 19:10

Mathematics, 03.11.2020 19:10


Mathematics, 03.11.2020 19:10

Mathematics, 03.11.2020 19:10

Mathematics, 03.11.2020 19:10


Social Studies, 03.11.2020 19:10


Mathematics, 03.11.2020 19:10

Social Studies, 03.11.2020 19:10

Mathematics, 03.11.2020 19:10

Mathematics, 03.11.2020 19:10