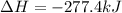Calculate ΔH o rxn for the following: CH4(g) + Cl2(g) → CCl4(l) + HCl(g)[unbalanced] ΔH o f [CH4(g)] = −74.87 kJ/mol ΔH o f [CCl4(g)] = −96.0 kJ/mol ΔH o f [CCl4(l)] = −139 kJ/mol ΔH o f [HCl(g)] = −92.31 kJ/mol ΔH o f [HCl(aq)] = −167.46 kJ/mol ΔH o f [Cl(g)] = 121.0 kJ/mol

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 21:20
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1

Chemistry, 23.06.2019 00:30
When did stem cell research become known ? who discovered stem cell? what experiments or studies have been conducted so far?
Answers: 3

Chemistry, 23.06.2019 03:00
Determine type of reaction & predict the product c3h12+o2 =
Answers: 1
You know the right answer?
Calculate ΔH o rxn for the following: CH4(g) + Cl2(g) → CCl4(l) + HCl(g)[unbalanced] ΔH o f [CH4(g)]...
Questions

Biology, 14.12.2020 08:50

History, 14.12.2020 08:50


Mathematics, 14.12.2020 08:50

Advanced Placement (AP), 14.12.2020 08:50

Social Studies, 14.12.2020 08:50

Mathematics, 14.12.2020 08:50

Mathematics, 14.12.2020 08:50


Computers and Technology, 14.12.2020 08:50

Mathematics, 14.12.2020 08:50

Chemistry, 14.12.2020 08:50


Mathematics, 14.12.2020 08:50



Social Studies, 14.12.2020 08:50


Mathematics, 14.12.2020 08:50

Biology, 14.12.2020 08:50


![\Delta H=\sum [n\times \Delta H(products)]-\sum [n\times \Delta H(reactant)]](/tpl/images/0550/2259/948cd.png)
![\Delta H=[(n_{CCl_4}\times \Delta H_{CCl_4})+(n_{HCl}\times B.E_{HCl}) ]-[(n_{CH_4}\times \Delta H_{CH_4})+n_{Cl_2}\times \Delta H_{Cl_2}]](/tpl/images/0550/2259/ca379.png)
![\Delta H=[(1\times -139)+(1\times -92.31) ]-[(1\times -74.87)+(1\times 121.0]](/tpl/images/0550/2259/0d24c.png)