
Chemistry, 17.03.2020 00:57 gracie6313
You take an aspirin tablet (a compound consisting solely of carbon, hydrogen, and oxygen) with a mass of 1.00 g, burn it in air, and collect 2.20 g of carbon dioxide and 0.400 g water. The molar mass of aspirin is between 170 and 190 g/mol. The molecular formula of aspirin is

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 22.06.2019 10:00
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2
You know the right answer?
You take an aspirin tablet (a compound consisting solely of carbon, hydrogen, and oxygen) with a mas...
Questions

Social Studies, 22.07.2019 14:50



Computers and Technology, 22.07.2019 14:50



Physics, 22.07.2019 14:50

Biology, 22.07.2019 14:50

Mathematics, 22.07.2019 14:50

Mathematics, 22.07.2019 14:50





History, 22.07.2019 14:50

Social Studies, 22.07.2019 14:50


Social Studies, 22.07.2019 14:50

History, 22.07.2019 14:50

Mathematics, 22.07.2019 14:50

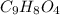
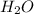 = 0.400 g /18 g/mol = 0.0222 moles
= 0.400 g /18 g/mol = 0.0222 moles
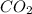 = 2.20 g /44.01 g/mol = 0.05 moles
= 2.20 g /44.01 g/mol = 0.05 moles


