
Chemistry, 17.03.2020 00:30 zachzach28280
Be sure to answer all parts. A buffer consists of 0.38 M KHCO3 and 0.71 M K2CO3. Carbonic acid is a diprotic acid with Ka1 = 4.5 × 10−7 and Ka2 = 4.7 × 10−11. (a) Which Ka value is more important to this buffer?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Perform the following mathematical operations and report the answer to the appropriate number of significant figures 5.87998 + 3.100
Answers: 2

Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3
You know the right answer?
Be sure to answer all parts. A buffer consists of 0.38 M KHCO3 and 0.71 M K2CO3. Carbonic acid is a...
Questions


Chemistry, 12.09.2019 18:30

Mathematics, 12.09.2019 18:30


Chemistry, 12.09.2019 18:30

Chemistry, 12.09.2019 18:30

History, 12.09.2019 18:30



History, 12.09.2019 18:30


History, 12.09.2019 18:30

History, 12.09.2019 18:30


Mathematics, 12.09.2019 18:30

English, 12.09.2019 18:30



History, 12.09.2019 18:30

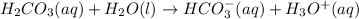
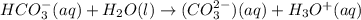
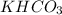 and
and 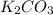 . This means that there will be
. This means that there will be 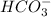 ions and
ions and 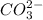 ions and since, both of them are present in the second step.
ions and since, both of them are present in the second step. 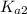 is more significant with respect to this reaction.
is more significant with respect to this reaction. 


