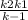Chemistry, 16.03.2020 23:27 kjmccarty02
The rate law for the decomposition of ozone to molecular oxygen is rate = k[O3]2 [O2] The mechanism proposed for this process is O3 0001 O + O2 O + O3 2O2 What is the rate law in terms of k1, k2, and k−1? Be sure to simplify the rate law before inputting your answer.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 14:50
Complete the following statements to describe solids, liquids, and gases. select the correct answer from each drop-down menu. a solid a definite volume and a definite shape. a liquid a definite volume and a definite shape. a gas a definite volume and a definite shape
Answers: 1

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 22.06.2019 20:30
Citric acid has a ph between 1 and 3. it is considered to be aa. weak acidb. weak basec. strong based. strong acid
Answers: 2
You know the right answer?
The rate law for the decomposition of ozone to molecular oxygen is rate = k[O3]2 [O2] The mechanism...
Questions





Mathematics, 29.01.2020 04:05



Mathematics, 29.01.2020 04:05

Social Studies, 29.01.2020 04:05

Mathematics, 29.01.2020 04:05


Physics, 29.01.2020 04:05




Mathematics, 29.01.2020 04:05


Mathematics, 29.01.2020 04:05


English, 29.01.2020 04:05

![k\frac{[O3]^{2} }{[O2]}\\](/tpl/images/0549/5602/5743c.png)
![\frac{k1 [O3]}{k-1[O2]}](/tpl/images/0549/5602/7e15c.png)
![k2 \frac{k1 [O3] [O3]}{k-1[O2]} = \frac{k2k1 [O3]^{2} }{k-1[O2]}\\](/tpl/images/0549/5602/a8827.png)