
Chemistry, 16.03.2020 17:31 Savannahh8503
The formation constant* of [M(CN) 4 ]2− is 7.70 × 10 16 , where M is a generic metal. A 0.150 mole quantity of M(NO3)2 is added to a liter of 0.820 M NaCN solution. What is the concentration of M2+ ions at equilibrium?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3


Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1
You know the right answer?
The formation constant* of [M(CN) 4 ]2− is 7.70 × 10 16 , where M is a generic metal. A 0.150 mole q...
Questions

Mathematics, 18.03.2021 15:40

Mathematics, 18.03.2021 15:40

Chemistry, 18.03.2021 15:40


Mathematics, 18.03.2021 15:40

Mathematics, 18.03.2021 15:40




History, 18.03.2021 15:40



Health, 18.03.2021 15:40


History, 18.03.2021 15:40

Physics, 18.03.2021 15:40

Mathematics, 18.03.2021 15:40


Mathematics, 18.03.2021 15:40

![M(NO_3)_2+NaCN\leftrightarrow [M(CN)_4]^{-2}+NaNO_3](/tpl/images/0548/6085/c1450.png)
![M^{+2}+4CN^-\leftrightarrow [M(CN)_4]^{-2}](/tpl/images/0548/6085/1b38d.png)
![K_F=\frac{[[M(CN)_4]^{-2}]_{eq}}{[M^{+2}]_{eq}[CN^{-}]_{eq}^4}](/tpl/images/0548/6085/aa364.png)
![[M^{+2}]_0=0.150M;[CN^-]_0=0.820M](/tpl/images/0548/6085/52301.png)
 due to the reaction progress:
due to the reaction progress: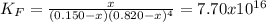
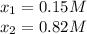
![[M^{+2}]_{eq}=0.15M-0.15M=0M](/tpl/images/0548/6085/e5b91.png)


