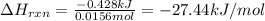Instant cold packs, often used to ice athletic injuries on the field, contain ammonium nitrate and water separated by a thin plastic divider. When the divider is broken, the ammonium nitrate dissolves according to the following endothermic reaction: NH4NO3(s)>NH+4(aq)+NO-3(aq) In order to measure the enthalpy change for this reaction, 1.25 g of NH4NO3 is dissolved in enough water to make 25.0 mL of solution. The initial temperature is 25.8 degree C and the final temperature (after the solid dissolves) is 21.9 degree C.
A. Calculate the change in enthalpy for the reaction in kilojoules per mole. (Use 1.0g/mL as the density of the solution and 4.18J/g degree C as the specific heat capacity.)
Express your answer to two significant figures and include the appropriate units.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Select each correct answer. more than one answer may be correct. which of the following is a characteristic of unicellular organisms? they can possess tissues and organs. all of their functions are performed by a single cell. they are usually microscopic. each of their cells is specialized to perform a specific function.
Answers: 1

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2
You know the right answer?
Instant cold packs, often used to ice athletic injuries on the field, contain ammonium nitrate and w...
Questions




Biology, 22.04.2020 23:43

Chemistry, 22.04.2020 23:43

Mathematics, 22.04.2020 23:43


History, 22.04.2020 23:43

Mathematics, 22.04.2020 23:43

Mathematics, 22.04.2020 23:43


Health, 22.04.2020 23:43




Advanced Placement (AP), 22.04.2020 23:44


English, 22.04.2020 23:44

World Languages, 22.04.2020 23:44

Mathematics, 22.04.2020 23:44

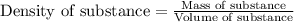
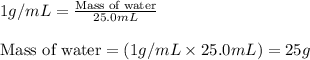
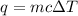
 = change in temperature =
= change in temperature = 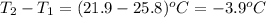
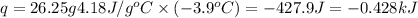
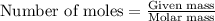
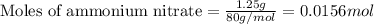
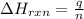
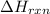 = enthalpy change of the reaction
= enthalpy change of the reaction