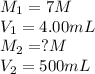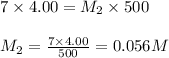Chemistry, 13.03.2020 19:16 bglosson4333
A 4.00 g mass of NaOH is dissolved in 5.00 mL of water. A 4.00-mL aliquot of this solution is then diluted to exactly 500 mL of solution using distilled water. What is molar concentration of NaOH in the final diluted solution?

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 05:30
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1

Chemistry, 22.06.2019 10:00
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2
You know the right answer?
A 4.00 g mass of NaOH is dissolved in 5.00 mL of water. A 4.00-mL aliquot of this solution is then d...
Questions

Mathematics, 25.05.2021 15:30



Mathematics, 25.05.2021 15:30

Mathematics, 25.05.2021 15:30

English, 25.05.2021 15:30


Social Studies, 25.05.2021 15:30




History, 25.05.2021 15:30


Chemistry, 25.05.2021 15:30


Physics, 25.05.2021 15:30




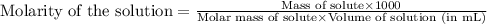
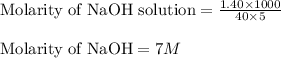
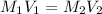
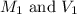 are the molarity and volume of the concentrated NaOH solution
are the molarity and volume of the concentrated NaOH solution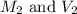 are the molarity and volume of diluted NaOH solution
are the molarity and volume of diluted NaOH solution