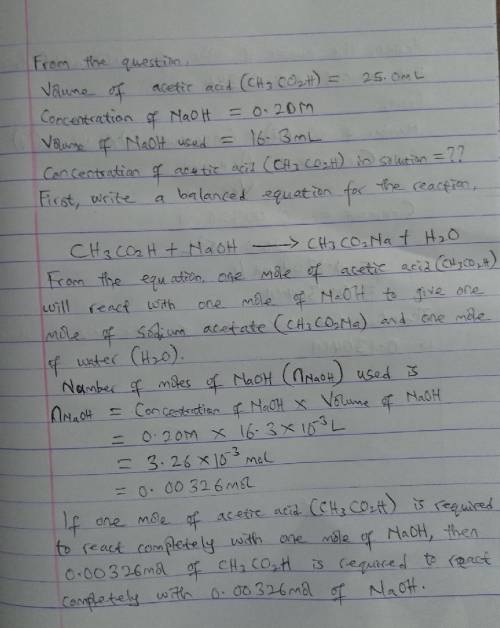Chemistry, 12.03.2020 06:08 JavyHart9695
A 25.0mL solution acetic acid (CH3CO2H) is titrated with 0.20M NaOH and reaches the endpoint after the addition of 16.3mL of NaOH. What is the concentration of acetic acid in solution

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:50
Which statement describes how phase changes can be diagrammed as a substance is heated? the phase is on the y-axis and the temperature is on the x-axis. the temperature is on the y-axis and the phase is on the x-axis. the time is on the y-axis and the temperature is on the x-axis. the temperature is on the y-axis and the time is on the x-axis.
Answers: 1


Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1
You know the right answer?
A 25.0mL solution acetic acid (CH3CO2H) is titrated with 0.20M NaOH and reaches the endpoint after t...
Questions

Mathematics, 24.11.2021 14:00


Mathematics, 24.11.2021 14:00


English, 24.11.2021 14:00



Social Studies, 24.11.2021 14:00

Biology, 24.11.2021 14:00


Social Studies, 24.11.2021 14:00

English, 24.11.2021 14:00


Mathematics, 24.11.2021 14:00


Mathematics, 24.11.2021 14:00