
Chemistry, 12.03.2020 02:42 natekotterman
Most of the sulfur used in the United States is chemically synthesized from hydrogen sulfide gas recovered from natural gas wells. In the first step of this synthesis, called the Claus process, hydrogen sulfide gas is reacted with dioxygen gas to produce gaseous sulfur dioxide and water.
Suppose a chemical engineer studying a new catalyst for the Claus reaction finds that 994 liters per second of dioxygen are consumed when the reaction is run at 170°C and 0.77 atm. Calculate the rate at which sulfur dioxide is being produced. Give your answer in kilograms per second. Round your answer to 2 significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1

Chemistry, 23.06.2019 00:30
What are the advantages of using the metric system? designed as a decimal system making conversions simpler more accurate system of measurement has prefixes that correspond to an amount to use with all base units used by the entire scientific community
Answers: 2

You know the right answer?
Most of the sulfur used in the United States is chemically synthesized from hydrogen sulfide gas rec...
Questions


Mathematics, 05.05.2020 19:06







History, 05.05.2020 19:06


Chemistry, 05.05.2020 19:06

Mathematics, 05.05.2020 19:06





English, 05.05.2020 19:06

Mathematics, 05.05.2020 19:06


English, 05.05.2020 19:06

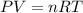 ( ideapl gas equation)
( ideapl gas equation)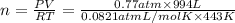
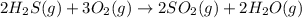 (Claus process)
(Claus process)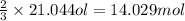 of sulfur dioxide
of sulfur dioxide

