
Chemistry, 11.03.2020 23:39 samantha9014
An aqueous potassium iodate ( KIO3 ) solution is made by dissolving 562 562 grams of KIO 3 KIO3 in sufficient water so that the final volume of the solution is 4.30 L. Calculate the molarity of the KIO 3 KIO3 solution.

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 03:20
High-pressure liquid chromatography (hplc) is a method used in chemistry and biochemistry to purify chemical substances. the pressures used in this procedure range from around 500 kilopascals (500,000 pa) to about 60,000 kpa (60,000,000 pa). it is often convenient to know the pressure in torr. if an hplc procedure is running at a pressure of 1.03×108 pa , what is its running pressure in torr?
Answers: 3

Chemistry, 23.06.2019 07:00
An unknown substance is a white solid at room temperature and has a melting point of 78 °c. which of the following substances is most likely to be the identity of the unknown sample? a. naphthalene, a molecular solid with the formula c10h8 b. silica, a network solid held together by covalent bonds with the formula sio2 c. calcium chloride, an ionic compound with the formula cacl2 d. water, an molecular compound with the formula h2o
Answers: 2

Chemistry, 23.06.2019 07:00
Agas has an empirical formula ch4. 0.16g of the gas occupies a volume of 240cm^3 what is the molecular formula of the me anyone who !
Answers: 1

Chemistry, 23.06.2019 07:20
Which statement explains which component is likely to be more powerful in explaining a scientific phenomenon? a) component c, because a theory is often passed on possibility and not certainty b) component d, because a hypothesis is often based on possibility not certainty c) component c, because the ability to explain several occurrences in the natural world is a characteristic of a hypothesis d) component d, because the ability to explain several occurrences in the natural world is a characteristic of a theory
Answers: 3
You know the right answer?
An aqueous potassium iodate ( KIO3 ) solution is made by dissolving 562 562 grams of KIO 3 KIO3 in s...
Questions



History, 05.05.2020 14:05

Biology, 05.05.2020 14:05

Mathematics, 05.05.2020 14:05

Advanced Placement (AP), 05.05.2020 14:05

Biology, 05.05.2020 14:05


Biology, 05.05.2020 14:05









Mathematics, 05.05.2020 14:06


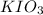 solution is 0.612 mol/L
solution is 0.612 mol/L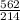 moles of
moles of 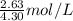 = 0.612 mol/L
= 0.612 mol/L

