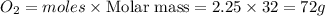Chemistry, 11.03.2020 18:36 aeshaalhemri
The combustion of propane may be described by the chemical equation C 3 H 8 ( g ) + 5 O 2 ( g ) ⟶ 3 CO 2 ( g ) + 4 H 2 O ( g ) C3H8(g)+5O2(g)⟶3CO2(g)+4H2O(g) How many grams of O 2 ( g ) O2(g) are needed to completely burn 19.7 g C 3 H 8 ( g ) ?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2
You know the right answer?
The combustion of propane may be described by the chemical equation C 3 H 8 ( g ) + 5 O 2 ( g ) ⟶ 3...
Questions



Mathematics, 03.01.2021 09:50



Chemistry, 03.01.2021 09:50


Chemistry, 03.01.2021 09:50

Biology, 03.01.2021 09:50



History, 03.01.2021 09:50



Mathematics, 03.01.2021 14:00


English, 03.01.2021 14:00

Biology, 03.01.2021 14:00

English, 03.01.2021 14:00

SAT, 03.01.2021 14:00

 are needed to completely burn 19.7 g
are needed to completely burn 19.7 g 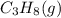
 of particles.
of particles.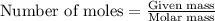
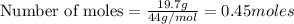
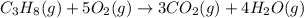
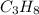 requires 5 moles of oxygen
requires 5 moles of oxygen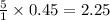 moles of oxygen
moles of oxygen