

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Activity two: just lemons, inc. production here's a one-batch sample of just lemons lemonade production. determine the percent yield and amount of leftover ingredients for lemonade production and place your answers in the data chart. hint: complete stoichiometry calculations for each ingredient to determine the theoretical yield. complete a limiting reactant-to-excess reactant calculation for both excess ingredients. water sugar lemon juice lemonade percent yield leftover ingredients 946.36 g 196.86 g 193.37 g 2050.25 g just lemons lemonade recipe equation: 2 water + sugar + lemon juice = 4 lemonade mole conversion factors: 1 mole of water = 1 cup = 236.59 g 1 mole of sugar = 1 cup = 225 g 1 mole of lemon juice = 1 cup = 257.83 g 1 mole of lemonade = 1 cup = 719.42 g
Answers: 2

Chemistry, 21.06.2019 19:00
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1

Chemistry, 21.06.2019 23:00
50 pts plz what is the physical state of matter of baking soda.
Answers: 1

You know the right answer?
Calculate the mass of H2O produced by metabolism of 1.2 kg of fat, assuming the fat consists entirel...
Questions



Mathematics, 01.04.2020 18:42


Mathematics, 01.04.2020 18:42

Mathematics, 01.04.2020 18:42





Physics, 01.04.2020 18:43



Mathematics, 01.04.2020 18:43

Mathematics, 01.04.2020 18:43



Mathematics, 01.04.2020 18:43

Mathematics, 01.04.2020 18:43

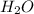 is produced
is produced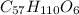 +
+ 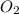 ⇒
⇒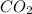 +
+ 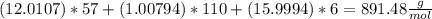
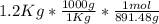
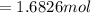
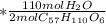
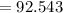 mol
mol 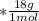 =1665.7 g
=1665.7 g 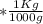 ≅ 1.7 Kg
≅ 1.7 Kg

