

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 20:00
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
What is the molar solubility of AgCl in 0.10 M NaCN if the colorless complex ion Ag(CN)2- forms? Ksp...
Questions

Mathematics, 04.10.2019 20:30



History, 04.10.2019 20:30

Social Studies, 04.10.2019 20:30

Mathematics, 04.10.2019 20:30

Mathematics, 04.10.2019 20:30

Mathematics, 04.10.2019 20:30

Social Studies, 04.10.2019 20:30


Mathematics, 04.10.2019 20:30


Mathematics, 04.10.2019 20:30


Mathematics, 04.10.2019 20:30

Physics, 04.10.2019 20:30

Biology, 04.10.2019 20:30

Computers and Technology, 04.10.2019 20:30

Social Studies, 04.10.2019 20:30

![Ksp = [Ag^{+}][Cl^{-}] = 1.8 \cdot 10^{-10}](/tpl/images/0539/7706/583a7.png) (1)
(1) ![Kf = \frac{[Ag(CN)_{2}^{-}]}{[Ag^{+}][CN^{-}]^{2}} = 1.0 \cdot 10^{21}](/tpl/images/0539/7706/daa6c.png) (2)
(2)![Keq = \frac{[Ag(CN)_{2}^{-})][Cl^{-}]}{[CN^{-}]^{2}}](/tpl/images/0539/7706/f78ba.png) (3)
(3)![Keq = \frac{[Ag(CN)_{2}^{-})]}{[CN^{-}]^{2}} \frac{Ksp}{[Ag^{+}]} = Kf \cdot Ksp = 1.0 \cdot 10^{21} \cdot 1.8 \cdot 10^{-10} = 1.8 \cdot 10^{11}](/tpl/images/0539/7706/79edb.png)
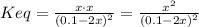 (4)
(4)

