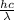Chemistry, 10.03.2020 01:34 tsmalls70988
The first ionization energy, E , of a nitrogen atom is 2.32 aJ. What is the wavelength of light, in nanometers, that is just sufficient to ionize a nitrogen atom? Values for constants can be found in the Chempendix.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 05:30
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 19:00
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1
You know the right answer?
The first ionization energy, E , of a nitrogen atom is 2.32 aJ. What is the wavelength of light, in...
Questions

Biology, 20.09.2019 00:30




History, 20.09.2019 00:30

History, 20.09.2019 00:30




English, 20.09.2019 00:30

Arts, 20.09.2019 00:30

Mathematics, 20.09.2019 00:30

Mathematics, 20.09.2019 00:30


Spanish, 20.09.2019 00:30





Mathematics, 20.09.2019 00:30