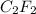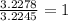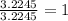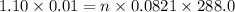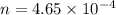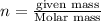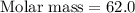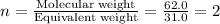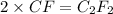Chemistry, 10.03.2020 00:29 jjimenez0276
A 0.02887 g sample of gas occupies 10.0 mL at 288.0 K and 1.10 atm. Upon further analysis, the compound is found to be 38.734 % C and 61.266 % F . What is the molecular formula of the compound?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3

Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1

Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 23.06.2019 00:10
Covalent compounds: mastery test select the correct answer what is formed when atoms join together with a covalent bond? a. an ion b. a molecule c. a neutral atom d. a noble gas
Answers: 3
You know the right answer?
A 0.02887 g sample of gas occupies 10.0 mL at 288.0 K and 1.10 atm. Upon further analysis, the compo...
Questions








Mathematics, 05.11.2019 22:31